 |
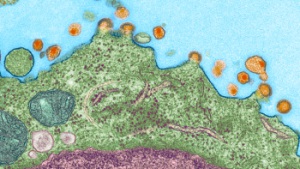
| In this image, captured by an electron microscope, HIV (orange) is budding off this CD4 T immune cells (green). Credit: Jinny Wong |
As it turns out, HIV is not guilty of murdering CD4 cells, rather of instigating cellular suicide.
The master sleuths responsible for this groundbreaking discovery hail from the laboratory of Warner C. Greene, MD, PhD, who is the director of virology and immunology research at Gladstone Institutes, a biomedical research nonprofit affiliated with the University of California, San Francisco (UCSF). Greene and his colleagues recently published simultaneous papers in Nature and in Science in which they identified how this suicide process works. After HIV attempts and fails to infect a resting CD4 cell—this is called an abortive infection—particles of the virus remain in the cell and are detected by a protein called IFI16. This protein then signals an enzyme known as caspase-1 to instigate a highly inflammatory death, known as pyroptosis, of these “abortively infected” resting CD4s. The inflammation attracts more CD4s to the “hot zone,” where they fall prey to this self-reinforcing cycle of destruction. Consequently, in the absence of antiretroviral (ARV) treatment, the immune system is shifted into suicidal overdrive, effectively depleting itself and causing AIDS.
“It’s been a very dynamic and thrilling scientific journey,” Greene says of his research. “Lab meetings have been quite fascinating. My jaw would drop at least once a month as a new result would come in and I would say, ‘Really?’”
Greene’s lab has also identified an existing drug called VX-765 that inhibits caspase-1 in tissue studies, preventing the enzyme from killing CD4s through pyroptosis and from leading to inflammation. Because the Vertex-owned drug has already been established as safe and well tolerated through Phase IIb trials for the treatment of epilepsy and psoriasis, the Gladstone researchers are hoping to jump into HIV-related Phase II human studies as soon as possible.
A persistent mystery in HIV research has been the fact that 95 percent of CD4 cells are in a resting state at any given time; and because HIV can only complete its life cycle and produce new virus if a CD4 cell is actively replicating, the virus cannot infect these resting cells. Nevertheless, these cells are still depleted by untreated HIV infection. Furthermore, having HIV infect and kill off the remaining 5 percent of CD4 cells—those that are actively replicating—is not enough of a hit to the overall immune system to cause the kind of devastation seen in advanced HIV disease.
“It’s always been an enigma here, because the number of infected cells was far less than the number of cells that died,” says Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases (NIAID), a division of the National Institutes of Health (NIH), who calls the Gladstone Institutes findings “an important advance.”
HIV attempts to infect the resting cells but fails at the step in the virus’ life cycle known as reverse transcription. Consequently, fragments of viral DNA remain in the abortively infected resting cell’s cytoplasm. This would cause the cell no harm were it not for the IFI16 protein, which, according to Greene’s research of spleen, tonsil and lymph-node tissue, then initiates the destructive chain reaction.
“The cell says, ‘Oh my God, I’m infected with something. Maybe I should die to protect the host,’” Greene says. “But in the case of HIV, what was originally designed as a protective response spins out of control. It leads to the death of CD4 T cells in unprecedented numbers.”
In this process of pyroptosis, the abortively infected CD4 cell releases inflammatory cytokines and its cytoplasmic content, which serve as a clarion call to the immune system. Greene theorizes that both active and resting CD4s are drawn to the site, leading to greater viral replication in active cells and abortive infection in the others.
By definition, inflammation occurs when cytokines and chemokines attract more white blood cells, such as CD4 cells, to the zone of inflammation. An acute case of inflammation is a good thing; it is how the body clears a bacterial or fungal infection, for example.
Actively infected cells perish more quietly than their abortively infected cousins: The caspase-3 enzyme kills the cell in a process known as apoptosis—which means “falling leaf,” while pyroptosis means “fiery fall.” In apoptosis, the CD4’s cytoplasm stays contained in the cell membranes and does not cause an inflammatory reaction. The membranes are then engulfed and digested by other immune cells called macrophages. Previous studies have suggested that this process is what causes AIDS; Greene’s research effectively refutes this notion. In a future paper, he will clarify why past research has been led astray.
One of the most profound elements of these news findings is that, for the first time, they link HIV’s two destructive signatures: chronic inflammation and depletion of the immune system. As the population of people living with HIV gets older, scientists believe that chronic inflammation—which occurs even in the context of successful ARV treatment—has led to the early development of common diseases associated with aging. Some of the results are higher rates of dementia, heart attack and cancer among people living with the virus.
“Most of the damage done by HIV can be inhibited by effective antiretroviral therapy,” says Steven Deeks, MD, a professor of medicine at UCSF who researches the role of chronic inflammation in people living with the virus. “For those who cannot access or tolerate these drugs, novel interventions aimed at preventing the harm of chronic inflammation might prove useful.”
One possibility is that, if cheap and widely available, a caspase-1 inhibitor such as VX-765 could serve as a stopgap measure to slow the progression of HIV disease among those in poorer nations who are awaiting treatment with triple antiretroviral therapy.
Pointing out that Greene’s research focused only on untreated tissue samples, Deeks says that there are theoretical reasons why inhibiting caspase-1 might still help HIV-positive people on ARVs, although not to the degree that it otherwise would in those who are not taking HIV therapy. “It might prove possible in the future to improve immune function and overall health for those having a suboptimal response to therapy,” he says
Also, because the inflammation that pyroptosis instigates spurs the proliferation of memory CD4 T cells, which are a part of the viral reservoir, Greene says that caspase-1 inhibitor might one day become a component of a multi-pronged HIV cure approach that slowly depletes the reservoir.
Calling such hopes “a bit of a stretch,” the NIAID’s Fauci nonetheless says, “I think that’s a possibility, but I don’t think that we should all get excited now that this is a real, giant step toward a cure. This is an important step toward understanding pathogenesis. Whether it’s an important step toward intervention is much too early to tell right now.”
While such a drug would have the benefit of working outside the viral life cycle and therefore would not be as likely to lead to drug resistance, there are potential drawbacks. Pyroptosis is integral to clearing various bacterial infections, so interrupting that natural process could lead those taking a caspase-1 inhibitor to become vulnerable to such infections.
“In the case of HIV infection I think that you will be saving CD4 T cells,” Greene says. “It will obviously be a therapeutic balance, as is every medication you give to someone.”
In order to solve these mysteries, Greene is focused on moving VX-765 into a clinical trial in humans. While using an existing compound that has already jumped over various regulatory hurdles allows for highly expedited research, he and his team still have many challenges ahead before they can begin such work. They must first come to an agreement with Vertex over use of the drug—they are currently in the midst of negotiations—and they also need to secure funding, whether that be from industry, governmental or private sources.
“If we had the money right now and they were willing to give us the drug,” Green says, “we easily could be in the clinic in six months.”
For more analysis, please read "Overlooked Caveats Regarding New CD4 T Cell Death Papers" by Richard Jefferys at Treatment Action Group (TAG).






3 Comments
3 Comments