UPDATE: On January 31, 2022, the Food and Drug Administration granted full approval of Moderna’s COVID-19 vaccine, to be marketed under the brand name Spikevax, for people ages 18 and older.
This article was originally published on December 18, 2020.
On December 18, the Food and Drug Administration (FDA) granted emergency use authorization for a second COVID-19 vaccine from Moderna and the National Institutes of Health. The emergency approval comes a day after an expert advisory panel voted almost unanimously that the vaccine’s benefits outweigh its risks.
“There is much we still do not know about SARS-CoV-2 and COVID-19. However, we do know that this vaccine is safe and can prevent symptomatic COVID-19 and severe disease,” National Institutes of Allergy and Infectious Disease director Anthony Fauci, MD, said in a statement. “It is my hope that all Americans will protect themselves by getting vaccinated when the vaccine becomes available to them. That is how our country will begin to heal and move forward.”
Today, FDA issued an emergency use authorization (EUA) for the second vaccine for the prevention of #COVID19 caused by SARS-CoV-2. The emergency use authorization allows the vaccine to be distributed in the U.S for use in individuals 18 years and older. https://t.co/w4BQVg0n2G pic.twitter.com/cw5JwYqxsh
— U.S. FDA (@US_FDA) December 19, 2020
Emergency use authorization is not the same as full approval, and it is typically granted after a simplified review. But given the political controversy around COVID-19 and the large number of healthy people who will receive the vaccine, the FDA undertook an extensive review process that included an outside expert advisory panel and public comment.
“With the availability of two vaccines now for the prevention of COVID-19, the FDA has taken another crucial step in the fight against this global pandemic that is causing vast numbers of hospitalizations and deaths in the United States each day,” FDA commissioner Stephen Hahn, MD, said in a press statement. “Through the FDA’s open and transparent scientific review process, two COVID-19 vaccines have been authorized in an expedited timeframe while adhering to the rigorous standards for safety, effectiveness and manufacturing quality needed to support emergency use authorization that the American people have come to expect from the FDA.”
A week ago, the FDA granted its first emergency use authorization for a COVID-19 vaccine from Pfizer and the German biotechnology company BioNTech. That vaccine is already being distributed throughout the country and given to health care workers and residents of long-term care facilities.
The next priority groups will include frontline essential workers who are at high risk for coronavirus exposure, people over age 65 and those with comorbidities, or underlying health conditions, that put them at greater risk for severe COVID-19 and death. States and local jurisdictions will make the final decisions about vaccine allocation.
Rapid rollout of a second vaccine will help address the dire shortage as COVID-19 cases surge in the United States. The number of COVID-19 deaths recently passed 300,000 in the U.S.—making it the leading cause of death—and approaches 1.7 million worldwide. Logistics will be simpler because the Moderna vaccine can be transported and stored at standard refrigerator temperatures (36° to 46° Fahrenheit), while the Pfizer/BioNTech vaccine requires an ultra-cold temperature (minus 94° Fahrenheit).
The Moderna vaccine was developed with support from Operation Warp Speed, and it will be distributed to Americans free of charge. The U.S. has contracted to buy 200 million doses of this vaccine, in addition to 100 million does of the Pfizer/BioNTech vaccine. Each recipient will need two doses.
Other experimental vaccines—including a chimpanzee adenovirus vector vaccine from AstraZeneca and the University of Oxford and a one-dose candidate from Johnson and Johnson—may also be authorized in the coming months.
Vaccine Effectiveness
On December 17, an FDA expert advisory committee voted 20 to 0, with one abstention, in favor of emergency authorization of the Moderna vaccine for people age 18 or older. The Pfizer/BioNTech vaccine is indicated for those age 16 and up, leading a few experts at least week’s committee meeting to vote against that vaccine due to limited data for people under 18.
Courtesy of National Institutes of Health
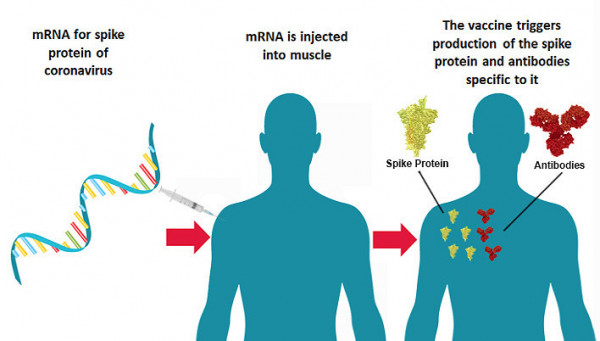
The Moderna vaccine (known as mRNA-1273), like the Pfizer/BioNTech vaccine, employs a novel messenger RNA (mRNA) approach. The vaccine uses lipid nanoparticles, or fat bubbles, to deliver bits of genetic material that encode instructions for making the SARS-CoV-2 spike protein—the red protuberances in the iconic virus image—which the coronavirus uses to enter human cells. When injected into a muscle, the cells produce the protein, triggering an immune response. The mRNA degrades quickly in the body and it does not alter human genes. The vaccines contain only fragments of viral genetic material, not the whole virus, so they cannot cause COVID-19.
No mRNA vaccines have been previously approved by the FDA, but the platform has been in development for many years. Once Chinese researchers revealed the genetic sequence of the new virus in January, Moderna was able to produce a vaccine candidate within a matter of days.
“Several years before SARS-CoV-2 entered the public consciousness, [National Institutes of Allergy and Infectious Diseases] scientists were working with Moderna to develop vaccines for other coronaviruses,” Fauci said. “That existing scientific foundation is what enabled both partners to move quickly to develop the mRNA-1273 vaccine candidate against the novel SARS-CoV-2 coronavirus.”
Early in the pandemic, Fauci predicted that a vaccine could be available in 12 to 18 months, but the two FDA authorizations beat even that optimistic timeline.
The Moderna vaccine was tested in a Phase III clinical trial that enrolled more than 30,000 volunteers. Men and women were about equally represented, and 25% were over age 65. One in five identified as Latino, 10% were Black and 5% were Asian. Nearly a quarter had comorbidities, including obesity, diabetes and chronic lung disease. More than 80% were considered at risk due to their occupation, including 25% who were health care workers. The study enrolled 176 people living with HIV.
Study participants were randomly assigned to receive two doses of the vaccine or saline placebo injections spaced four weeks apart.
As described a briefing document prepared by the FDA and another submitted by Moderna, the vaccine was 94.1% effective in reducing the risk of symptomatic COVID-19. A total of 196 cases of symptomatic COVID-19 were observed at least 14 days after the second dose: 185 in the placebo group and 11 in the vaccine group. In the main analysis, all 30 reported cases of severe COVID-19 occurred in the placebo group; one later severe case in the vaccine group awaits confirmation.
The vaccine was effective across all demographic groups. Effectiveness was high, at 86.4%, in people over age 65—an important consideration given that older people are at greatest risk for severe COVID-19 but they tend to mount a weaker immune response to vaccines.
What’s more, Moderna presented additional data indicating that the vaccine also appears to lower the risk of asymptomatic infection by two thirds. Reducing infection and transmission will be critical to halting the spread of the coronavirus and enabling life to return to—or at least towards—normal.
It is not yet known how long vaccine-induced immunity to SARS-CoV-2—or natural immunity after an infection—will last. However, earlier studies showed that this and other COVID-19 vaccines generate T-cell responses, which could persist even if antibody levels wane over time.
Most experts expect that so-called herd immunity could occur after around 70% of people have been vaccinated. At that point, there would be too few susceptible people for the virus to spread easily, and even people who are not vaccinated would be protected.
Vaccine Safety
The Moderna vaccine was safe and generally well tolerated, though many participants experienced mild to moderate side effects, especially after the second shot. Most vaccine recipients reported injection site reactions such as soreness. Other common symptoms included fatigue, headache, muscle and joint pain, chills, fever and swollen lymph nodes in the injected arm. Flu-like symptoms are not unusual after receiving vaccines and are an indication that the immune system is working. Three people developed Bell’s palsy, a sudden and usually temporary weakness or paralysis of facial muscles, which also occurred rarely in the Pfizer/BioNTech trial.
Shortly after the rollout of the Pfizer/BioNTech vaccine, two health care workers in the United Kingdom and three in Alaska experienced allergic reactions after vaccination. One did not have a prior history of such reactions. The Moderna vaccine uses similar technology. The reactions may have been triggered by polyethylene glycol, a stabilizer widely used in medical products, that is a component of both vaccines.
Responding to concerns, the FDA and Centers for Disease Control and Prevention (CDC) have indicated that these rare reactions should not derail the rollout of the vaccines. However, people with a history of vaccine reactions should discuss the risks with their providers. According to the CDC, a clinical provider with access to emergency supplies—including epinephrine and Benadryl—should be immediately available to assess and manage anaphylactic reactions. People with a history of such reactions should be observed for 30 minutes post-vaccination and others for 15 minutes.
People previously infected with SARS-CoV-2, many of whom are unaware of it because they were asymptomatic or could not get tested, can safely receive the vaccine, and some experts think it will probably offer additional protection.
Vaccine response and safety have not been analyzed separately for HIV-positive people in the Pfizer/BioNTech and Moderna trials. Outcomes also have not yet been determined for people with immune suppression, autoimmune conditions or cancer. The vaccine is not contraindicated for such individuals—and experts do not foresee problems—but they should talk to their care providers about their specific situation.
“There is no reason to believe that people with HIV should not get the vaccine. It is not a live vaccine and it is safe and efficacious across diverse groups,” Monica Gandhi, MD, MPH, medical director of the Ward 86 HIV clinic at Zuckerberg San Francisco General Hospital, told COVID Health, referring to both authorized vaccines. “I totally recommend it.”
The vaccines have not yet been studied in children or adolescents. A trial for those ages 12 to 17 is just getting underway, and a study for younger children is expected to start early next year.
The vaccines also have not yet been studied in pregnant people—an important consideration given that three quarters of health care workers are women, and a large proportion of them are of child-bearing age. Here, too, experts say that pregnant people should not be excluded from vaccination if they are otherwise eligible.
But persuading the population that the benefits of vaccination outweigh the risks could be a challenge. Some people are concerned that vaccines have been rushed through the development process due to political pressure. Others still do not regard COVID-19 as a serious threat. Polls suggest that African Americans and Latinos, who have higher rates of coronavirus infection and COVID-19 complications, may be less willing to get the vaccines.
Government and health officials have said that most health care workers should be vaccinated by the end of the year. Rollout for frontline workers, people over 65 and people with underlying conditions should be underway by the first months of 2021. Adults under age 65 with no particular risk factors could start getting vaccinated around April. Vaccination of this group could be completed between the summer and the end of next year, depending on willingness and an adequate supply.
Until then, it is still important to keep up precautions to prevent COVID-19, such as wearing masks and social distancing.
“The emergency use authorization of a second COVID-19 vaccine today means that millions more people can be immunized against this virus in the weeks to come,” Barbara Alexander, MD, president of the Infectious Diseases Society of America, said in a statement. “We should be proud of this remarkable accomplishment and we should make the most of this incredible scientific milestone. With strong federal funding for swift vaccine rollout and education, and by following the guidance of public health experts to continue wearing masks and maintaining distance, we can bring the end of this pandemic closer.”
Click here for more news about COVID-19 vaccines.






Comments
Comments