The annual Conference on Retroviruses and Opportunistic Infections (CROI) brings together thousands of the brightest minds in HIV science for a riveting confab about myriad efforts to combat the virus. For this year’s CROI, held February 13 to 16, the HIV research community descended upon the at times strangely sunny Seattle to share exciting information about, most notably, improved viral suppression rates, falling transmission rates and ever-more-advanced treatments for the virus.
Hepatitis C virus (HCV) carves out a significant niche at the conference as well. Click here to read an overview of CROI’s HCV-related presentations, many of which concern HIV/HCV coinfection. These studies also cover hep C–associated diseases, HCV testing, direct-acting antiviral cure rates, the benefits of treatment and sexual transmission of the virus among men who have sex with men (MSM).
Otherwise, keep reading for HIV-specific highlights from CROI 2017. To read more about any particular study, click the hyperlinks. You can also check out POZ’s newsfeed of articles about individual HIV-related studies and our sibling publication Hep’s newsfeed of HCV-related studies presented at the conference.

Demetre Daskalakis of the New York City Department of Health at CROI 2017 in Seattle, presenting exciting findings about progress in tackling New York City’s epidemic: http://www.croiwebcasts.org/console/player/33560?mediaType=slideVideo&Ben Ryan
Infection rates
The Centers for Disease Control and Prevention (CDC) provided conferencegoers some good news about improving HIV rates in the United States. After two decades of stagnation, the CDC’s estimate of the annual rate of new infections finally declined 18 percent between 2008 and 2014, from 45,700 to 37,600 per year.
Infection rates fell among all major risk groups during this time except for MSM, who saw a leveling off of new annual transmissions—which nonetheless is a sign of significant progress in bending the curve against the virus. Between 2008 and 2014, infection rates declined 56 percent among injection drug users (IDUs) and 36 percent among heterosexuals.
The estimated HIV rate for MSM remained at about 26,000 per year throughout this period, with rates holding steady among Blacks (at about 10,000 per year), dropping 18 percent among whites and rising 20 percent among Latinos.

Courtesy of CDC
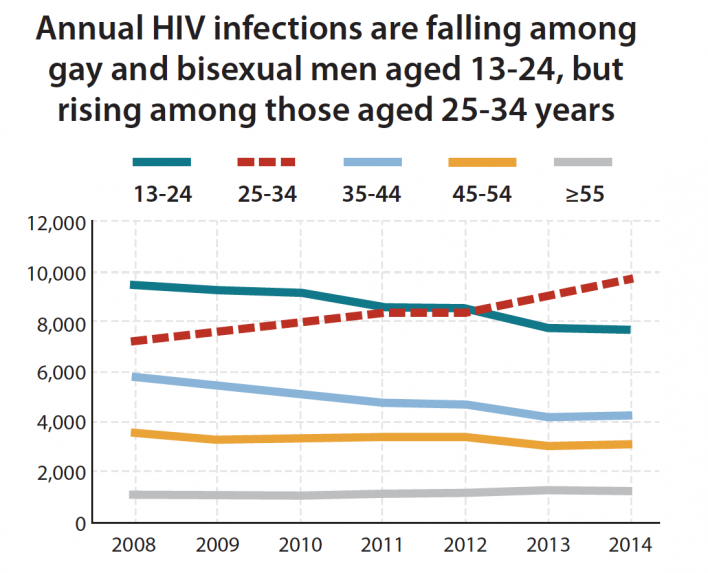
Infection trends among MSM varied by age, with 25- to 34-olds seeing a rise.Courtesy of CDC
Treatment
The HIV treatment pipeline shows promise for exciting advancements to come as clinical trials on new therapies and regimens advance.
The two-drug regimen of ViiV Healthcare’s Tivicay (dolutegravir) and Janssen’s Edurant (rilpivirine) suppressed HIV as well as standard three- or four-drug regimens at the 48-week point of an ongoing Phase III trial. (Clinical trials must complete three phases before a drug can be approved.
Merck’s investigational non-nucleoside reverse transcriptase inhibitor (NNRTI) doravirine suppressed HIV as well as and was better for cholesterol levels than Norvir (ritonavir)-boosted Prezista (darunavir) when either drug was combined with either Truvada or Epzicom (abacavir/lamivudine) in an ongoing Phase III trial.
Additionally, in a Phase II trial, two regimens, Descovy (emtricitabine/tenofovir alafenamide) plus Gilead Sciences’ investigational integrase inhibitor bictegravir or Tivicay, each suppressed HIV at high rates after 48 weeks in a Phase II trial.
Gilead also released 114-week results of its Phase III trial comparing Genvoya (elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide) with Stribild. (Genvoya, approved in November 2015, has the same components as Stribild but with an updated version of tenofovir that is safer for the bones and kidneys.) Genvoya actually did a better job suppressing HIV than Stribild and was also associated with a lower rate of discontinuation.
Last, in an ongoing Phase III study of people who had drug-resistant virus and very limited HIV treatment options, adding biweekly infusions of the monoclonal antibody ibalizumab to a standard HIV treatment regimen brought nearly half to full viral suppression by the 24-week point.

Paul E. Sax of Harvard Medical School at CROI 2017, presenting findings from the bictegravir study.Courtesy of Benjamin Ryan
PrEP
There was good news for women in a couple of PrEP studies. One study found that the effectiveness of oral PrEP in the Partners PrEP study, which gave at-risk HIV-negative women Truvada, Viread (tenofovir disoproxil fumarate) or a placebo, was unaffected by abnormal microbial states in their vaginas. Another found that the investigational vaginal ring form of PrEP does not compromise the effectiveness of hormonal birth control.
A New York City study found that a brief counseling session can boost adherence to PrEP, although follow-up sessions may be needed to ensure that the benefit endures.
Research of Seattle MSM starting PrEP found they had a very high rate of STIs, although evidence was inconclusive as to whether beginning PrEP affected STI rates. Meanwhile, French researchers found that if they gave a supply of the antibiotic doxycycline to men on PrEP with instructions to take a dose following condomless intercourse, it lowered their risk of chlamydia and syphilis but not gonorrhea.
With the Phase III trial of long-acting injectable cabotegravir as PrEP now getting under way, researchers recently conducted tests in monkeys and found that, at least in primates, those who were given this form of PrEP soon after infection with SIV, HIV’s simian cousin, were at risk of developing drug resistance.
Last, but certainly not least, was the case study of the Dutch man who according to numerous sources of evidence, adhered well to his daily PrEP regimen but contracted HIV nevertheless. This is the third documented case of PrEP failure and the first in which the individual did not contract a drug-resistant strain of the virus. Researchers speculated that his “remarkably high” number of anal sex partners—he reported between 38 and 75 per month—may have left him repeatedly exposed to HIV in such a way that he beat the odds, so to speak.
Elske Hoornenborg of the Public Health Service Amsterdam with her poster presentation about the PrEP failure case at CROI 2017Ben Ryan
Functional Cure
In a trial of 15 people who started antiretroviral (ARV) treatment soon after contracting HIV, researchers gave them multiple doses of the HIV Conserv vaccine and the drug romidepsin. After going off ARVs, five of 13 participants who have interrupted their ARVs, or 38 percent of the overall group, have been able to largely control the virus without a major viral rebound and without any other treatment for up to 28 weeks thus far.
Another study found that treating HIV in infants within eight days of birth has the potential to rapidly shrink the size of their reservoir of latently infected (unreplicating) cells. The persistence of the viral reservoir is what prevents standard ARV treatment from curing the virus, although in rare cases, very early treatment seems to have led to extended periods of viral remission—in one infant and one child in particular and in a handful of adults.

Istock
Viral Suppression
In an analysis of viral suppression rates among U.S. residents living with diagnosed HIV, CDC researchers found that about half of them had a fully suppressed viral load throughout 2014. Blacks, women, non-MSM (including injection drug users, or IDUs), those older than 55 and those diagnosed prior to 2013 were less likely to have durable viral suppression compared with other races, men, MSM, those younger than 55 and those diagnosed in 2013.
Another CDC study found that the overall amount of time that people receiving care for HIV in the United States spend with a viral load above 1,500 has fallen by about three quarters since the turn of the millennium. This likely helps explain the recent drop in HIV infection rates, since a viral load above 1,500 is considered significantly infectious.
The conference heard news that host city Seattle was among the first major U.S. metropolitan areas to hit the Joint United Nations Programme on HIV/AIDS’s (UNAIDS) so-called 90-90-90 targets, with at least 90 percent of its HIV population diagnosed, 90 percent of the diagnosed population on ARVs and 90 percent of those on ARVs virally suppressed. Meanwhile, a study in sub-Saharan Africa found that Zimbabwe, Malawi and Zambia are making excellent progress toward hitting the UNAIDS targets.

Istock
Cognitive Function
Two studies looked at abnormalities in the brain among people with HIV to try to get a better handle on the elevated rate of cognitive dysfunction seen in the HIV population. One study found that even when people with HIV are doing well on ARVs, they are more likely to have certain abnormalities in blood vessels in the brain that are associated with cognitive decline. That study was limited by the fact that it did not follow participants over time, so its authors could not speculate about how HIV may accelerate brain aging.
The other study did just that and followed people on successful ARV treatment over a two-year period. The investigators found that HIV was associated with abnormalities in brain structure and function but did not see any excess decline in these measures over time among the HIV-positive participants compared with those in an HIV-negative control group.

Istock
Drug Use Trends
A Seattle study found that rising injection use of meth may facilitated HIV’s spread among IDUs who are not MSM, who likely thanks to effective local syringe services programs currently have a low HIV rate. Researchers are concerned that non-MSM meth injectors report sharing injection materials with MSM, who have a very high HIV rate.
According to the CDC, the recreational use of opioids is as common as crystal meth among MSM in the United States. Looking at responses to survey questions about non–injection drug use provided in 2008, 2011 and 2014, CDC researchers found that the proportion reporting cocaine use held steady at about 17 to 19 percent; opioid use was also stable, at just below 8 percent, while meth use ranged between 6.6 and 8 percent.
Another CDC study found that nationwide, increasing proportions of those who started to inject drugs in the years since the mid-1990s first got hooked on opiate painkillers. This finding adds to a large body of evidence that an initial epidemic of addiction to pills like Vicodin and OxyContin has spilled over into an epidemic of addiction to injectable opioids, including heroin.

Thinkstock







Comments
Comments