HIV cure researchers received some disappointing news at the International AIDS Conference in Amsterdam (AIDS 2018), held in July. Two studies in particular offered a sobering reminder of how extraordinarily complex developing a safe and effective cure for the virus will be. More practically, investigators seek to induce so-called posttreatment control of HIV, or viral remission, in which a particular therapy would not eradicate the virus from the body but rather suppress HIV over an extended period without the need for long-term antiretroviral (ARV) treatment.
As conference attendees learned, the largest randomized trial of an HIV cure method recently failed to reduce viral DNA in human participants. The trial, called RIVER, tested the “kick and kill” strategy of seeking to roust from their slumber and then kill off latently infected immune cells that, because they don’t harbor actively replicating virus, remain under the radar of standard ARV treatment. (ARVs work only on cells that have replicating virus.) Such resting infected cells are a chief component of what is known as the viral reservoir, the stubborn persistence of which greatly frustrates cure efforts.

AmsterdamIstock
In a second study presented at the conference, an antibody treatment that had shown promise in monkeys failed to prompt posttreatment control of the virus after HIV-positive humans interrupted their ARV therapy.
Taking the pulse of her fellow HIV cure researchers, Sharon R. Lewin, MD, PhD, director of The Peter Doherty Institute for Infection and Immunity at the University of Melbourne in Australia, says, “If anything, there was probably more optimism four years ago because we had tried fewer things. We now know that curing HIV is definitely not an easy task.”
On the bright side, Lewin highlights other promising recent cure studies conducted in primates, noting, “We definitely have been able to cure a few monkeys. That’s exciting.”
But as the antibody study presented at the Amsterdam conference indicated, disappointing outcomes among humans may follow success in primate research.

Istock
“The preclinical studies have universally shown more favorable outcomes than human studies,” says Jintanat Ananworanich, MD, PhD, who in her capacity as the associate director for therapeutics research at the U.S. Military HIV Research Program directs research in the HIV cure field.“Although no strategies have resulted in remission in clinical trials thus far, tremendous knowledge on HIV persistence and immune responses has been generated. This is important to informing future trials.”
Concerns about recent setbacks notwithstanding, Lewin remains optimistic about the future of HIV cure research. “Science can also take dramatic turns with significant discoveries too,” she says. “So you never know what may change the field dramatically.”
Lewin is the lead author of a literature review recently published in The Lancet HIV, “Barriers and Strategies to Achieve a Cure for HIV,” in which she and her three coauthors offer a comprehensive summary of the impressive number of avenues researchers are pursuing in their quest for a cure, or something close to it. To follow are the main takeaways from their paper, as well as mentions of a few HIV cure studies published more recently.
Promising results raise hope
As Lewin and her colleagues write, the only person ever cured of HIV remains Timothy Ray Brown. As a component of his treatment for leukemia, Brown received stem cell transplants a decade ago from a donor with a genetic mutation that confers natural resistance to the virus—the surface of the donor’s immune cells lacked the CCR5 coreceptor to which most HIV attaches in order to infect the cells. As far as researchers can tell, Brown benefited from a sterilizing cure. There is no evidence in his body of any virus with the capacity to replicate, and his viral load has never rebounded.
Otherwise, in the realm of posttreatment control of HIV, quite a few people with the virus have been able to suppress their viral load for long stretches, sometimes for years, after interrupting standard ARV treatment. A recently published paper found that those who began ARVs very soon after contracting the virus are more likely to achieve such a prolonged state of viral remission after eventually going off their meds. It is likely that beginning on ARVs so promptly after infection keeps the viral reservoir relatively small, thus reducing the likelihood of latently infected cells springing to life at any given moment following a treatment interruption.
One of the most famous cases of such posttreatment control is that of the African child who was treated for HIV for less than a year after birth and, by the time his case was reported in 2017, had spent over eight years in a state of viral remission. In 2015, news surfaced that an 18-year-old French individual had spent 12 years off ARVs and still controlled the virus. Then, of course, there was the 2013 case of the Mississippi Child—met with great fanfare—who spent a couple of years off ARVs during her very young life but ultimately, and disappointingly, experienced a viral rebound at 4 years old.
Istock
According to Lewin, scientists’ increasingly enriched comprehension of the posttreatment-control phenomenon has made designing HIV cure studies more difficult. Researchers must take into account that some study participants might achieve control of their virus, even if for a short time, without the benefit of an investigative cure therapy, thus making it more challenging to prove that a cure treatment was the cause of viral remission or a delayed viral rebound after the interruption of ARV treatment.
What HIV cure researchers are up against
Not only do latently infected immune cells evade ARV treatment, but also for every million cells, perhaps only 60 harbor virus that can actually replicate; the rest of the latently infected cells contain defective virus. So finding those resting cells capable of waking up and repopulating the body with new virus in the absence of ARV treatment can be akin to finding a needle in a haystack. The immune system itself wastes considerable energy going after cells infected with dud copies of the virus.
In another of the myriad wily tricks HIV employs to ensure it sustains a lifelong infection, latently infected cells have the ability to clone themselves. Perhaps more than half of the viral reservoir cells in some people living with the virus are clones.
The matter of whether HIV continues to replicate at low levels in the face of effective ARV treatment has been the source of significant controversy in the cure field. A study presented at the 2018 Conference on Retroviruses and Opportunistic Infections (CROI) in Boston found no evidence of such ongoing replication in the lymph nodes, calling into question the notable contrasting findings of a 2016 paper.

The National Cancer Institute’s Mary Kearny, PhD, speaks at CROI 2018 in Boston about her study on whether fully suppressed HIV replicates in the lymph nodes.Benjamin Ryan
The challenge of measurement
The lack of precise tests for measuring the viral reservoir remains a considerable obstacle for HIV researchers. Currently, scientists in the field must rely on rather crude metrics, such as changes in the overall presence of viral DNA or RNA in the blood, to gauge how a particular treatment affects the size of the reservoir. (HIV carries its genetic code in RNA, which is copied to DNA following infection of a cell.) Such measures can underestimate the population of infected cells because most virus hides in tissues, not blood.
Scientists may also attempt to measure success by determining whether an HIV cure treatment is associated with a delay in viral rebound after ARV therapy is interrupted and whether such an intervention is associated with a particular level of control of the virus in the absence of ARVs.
If only scientists could identify a specific biomarker, such as a specific protein, that could predict the likelihood of a delay to viral rebound or control of the virus after a treatment interruption, study participants might be spared the burden of interrupting their ARVs, an increasingly common requirement in HIV cure study designs. Asking people to stop standard HIV treatment raises ethical questions and may discourage people living with the virus from entering cure trials. That said, multiple studies have indicated that treatment interruptions in cure studies are, in fact, safe.
Lewin argues that such a tidy biomarker would likely attract greater investment in cure research from pharmaceutical companies. (Currently, international funding into HIV cure research comes largely from private or governmental sources, mostly from the National Institutes of Health.) Because such for-profit companies prefer, for one, study designs boasting a level of simplicity that will help an investigational treatment pass muster with regulatory bodies like the Food and Drug Administration, and, two, to spend their research and development dollars efficiently, their researchers prefer clearly delineated, objective means of measuring success in clinical trials of experimental agents.

Total global funding for HIV cure research—this bar graph represents a tally of all sources—has surged in recent years, driven primarily by the NIH’s increasing investment in the field, according to AVAC.AVAC
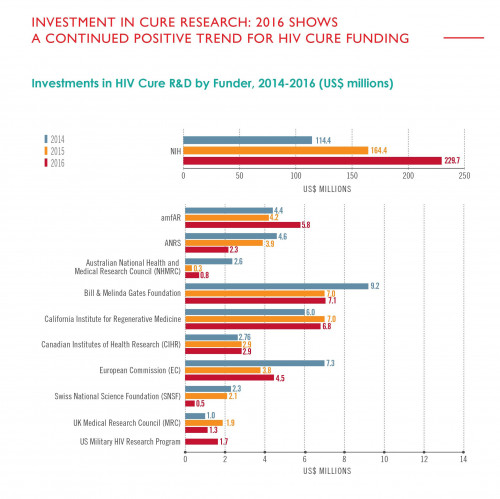
Included in this chart are all global funders who invested $1 million or more in HIV cure research during at least one year between 2014 and 2016, according to AVAC.AVAC
Case in point: The recent discovery of a biomarker that can predict if an individual will achieve a functional cure of hepatitis B virus (HBV) gave rise to a surge of interest from the pharmaceutical industry in researching curative therapies for HBV.
The various HIV cure methods currently under investigation:
Stem cell transplants
Clinicians have yet to replicate the success of Timothy Brown’s HIV cure. Multiple other individuals with cancer have since received stem cell transplants from donors who also have the genetic mutation related to the CCR5 coreceptor that confers resistance to the virus. One of the six such individuals whose cases have been published in scientific literature experienced a viral rebound (POZ interviewed him in 2015); the other five ultimately died as a result of complications following their stem cell transplant or from their underlying cancer.
In other cases of people living with HIV who received a stem cell transplant but from a donor who lacked the CCR5-related genetic mutation, the stem cell transplant did indeed delay the time to viral rebound, by 3 to 10 months, after the individuals stopped ARVs. However, the high fatality rate following transplantation highlights how impractical, not to mention unethical, this method of attempting to cure HIV is for anyone not already facing a high risk of death due to cancer.
Gene therapy
Seeking safer alternatives to cancer-treatment-based stem cell transplants, researchers are experimenting with gene-editing techniques to alter the DNA of an individual’s immune cells. In particular, the scientists will introduce genetic changes to deactivate the gene that gives rise to the CCR5 coreceptor, thus robbing HIV of a means of latching on to immune cells. The modified cells are then grown outside the body and ultimately reinfused into the person’s body. The aim is to spawn a population of immune cells that are resistant to infection. Hopefully, as the field of gene editing rapidly evolves, new, even more cutting-edge technology will facilitate progress on the HIV cure front.
Kick and kill
The method of waking up latently infected cells (the “kick” part) and then finishing them off (the “kill” part) has yielded some notably disappointing results of late, including those of the RIVER study that was presented at the conference in Amsterdam in July. Researchers pursuing this strategy have looked to various cancer drugs known as HDAC inhibitors as the kick element; but thus far, they have not been able to show such drugs can lead to a kill of reservoir cells.
Lewin remains cautiously optimistic about further research into these medications, noting that the RIVER trial used a less advanced and relatively weak kick agent. Recent, more preliminary studies that have examined other kick agents, such as so-called TLR agonists, have shown far greater promise.
On this front, Gilead Sciences, for one, is investigating a drug known as GS-9620 that has shown positive results in primate research.
Latency silencing: “block and lock”
Effectively the opposite of the kick and kill strategy, the “block and lock” method, also known as latency silencing, is based on the presumption that if rooting out and killing all the latently infected cells in the body is too challenging, keeping them in a silent state indefinitely may be a viable alternative. A recent study conducted in mice sought to inhibit a viral protein known as tat that acts as an on-off switch for viral replication in cells. The study successfully reduced the amount of HIV RNA expressed in tissue biopsies from the animals and delayed viral rebound after the interruption of ARV treatment.

Istock
Enhancing the immune system
Researchers are investigating using vaccines as a tool to prompt the body to better control the virus.
Scientists have also invested considerable energy into studying so-called broadly neutralizing antibodies, which are natural antibodies with the capacity to combat a wide array of HIV strains. Research has indicated that some of these antibodies are associated with a delay in viral rebound after an ARV treatment interruption. Recently, scientists have gone high-tech by synthesizing three such antibodies into one “trispecific” antibody—a kind of all-in-one triple combination therapy—that has already shown promise in its use as pre-exposure prophylaxis (PrEP) among primates.
Modulating the immune system
Scientists are seeking to manipulate proteins that redirect the traffic of immune fighting cells. One such example is an antibody called vedolizumab that targets a protein on the surface of CD4 cells and stops these cells from moving into the gut, where HIV focuses much of its assault on the immune system. An initial study in monkeys reported two years ago provided hope for progress in this area of research, but scientists recently repeated the study and found that the antibody had a null results on the second go-round. Preliminary results in humans also showed that vedolizumab had no effect on the time to viral rebound after individuals interrupted their ARV treatment.
Looking to the future
In all likelihood, a successful HIV cure, or posttreatment control, strategy will rely upon a combination approach based on a number of the methods currently under investigation or those yet to be imagined.

Dreamstime
“It is clear that achieving HIV remission will not be easy and that one should not expect any single intervention to help people get to remission,” says Jintanat Ananworanich.“We are taking small steps in discovery science.”
Any successful method will need to be safe, effective and, if it is to make a significant dent in the global epidemic, scalable. An HIV cure therapy that is extraordinarily expensive thanks to, for example, the highly involved and complex process required to provide personally tailored genetic editing of an individual’s immune cells, will have little to offer poorer nations—in particular those in sub-Saharan Africa—where the need is greatest.
Curative hepatitis C virus (HCV) treatment, for example, costs tens of thousands of dollars in the United States, which has led insurers to restrict coverage of the medications. The actual cost to manufacture such medications, however, is relatively low, which allows for a steep sliding scale elsewhere around the world.
The future of HIV cure science is also up against the phenomenal success of ARV treatment, which has set a high bar for any alternative means of suppressing the virus. Not only is the life expectancy of those on ARVs approaching normal but maintaining a fully suppressed viral load is associated with effectively zero risk of transmission.
However, such benefits don’t speak to the psychic costs of living with a highly stigmatized lifelong infection or how a cure therapy may alleviate such burdens. Then there is the extreme difficulty and expense of getting the global population on lifelong ARV treatment. Also, even well treated HIV is associated with an increased risk of numerous health conditions, such as cardiovascular disease and cognitive decline.
Some form of HIV cure could help address these problems. On the other hand, as HIV drug development continues to progress and long-acting injectable treatments, or even very long-lasting implants, become the standard of care, HIV cure treatments may cease to offer the freedom daily medications as an advantage over standard ARV therapy. (Or perhaps by then, long-acting antibody treatments will be the norm.)
Furthermore, if someone is in a state of posttreatment control of the virus, what reassurances will there be that the virus will remain dormant indefinitely and won’t suddenly surge back and make an individual unwittingly infectious? How frequently will people in a state of viral remission need viral load monitoring—and at what cost?
These pressing questions, along with HIV’s extraordinary complexity, likely make for a long and winding scientific road ahead. But thanks to the increasing funds backing such research and a growing army of top-tier scientists doggedly pursuing a cure, the future will hopefully prove bright with new developments.
Still, this field isn’t simply concerned with a binary outcome of finding the holy grail of a cure or otherwise failing to do so. Success will likely prove incremental, with scientists eventually discovering new means of further mitigating HIV’s long-term harms, further transforming a once surely fatal infection into an increasingly innocuous presence in the body and around the world.
Benjamin Ryan is POZ’s editor at large, responsible for HIV science reporting. His work has also appeared in The New York Times, New York, The Nation, The Atlantic and The Marshall Project. Follow him on Facebook, Twitter and on his website, benryan.net.






4 Comments
4 Comments