The late aughts were a vexing time for HIV prevention in the United States. The Centers for Disease Control and Prevention (CDC) estimated that the virus’s overall annual transmission rate had remained stagnant for nearly two decades. Diagnosis rates were rising among men who have sex with men (MSM), who had become increasingly lax about condom use since the introduction of effective antiretroviral (ARV) treatment for HIV ended the crisis years of the epidemic in 1996.
It was high time for a game changer.
Along came what is known as biomedical prevention of HIV. During the 2010s, HIV prevention science has been dominated by the fact, proven with increasing clarity by a series of important studies, that the medications used to treat the virus have a powerful capacity to prevent its spread. To wit, HIV-positive individuals who maintain an undetectable viral load thanks to ARVs have an extremely low risk of passing the virus to others; the risk may in fact be zero.
And then there’s pre-exposure prophylaxis (PrEP). In 2010, researchers published a landmark global study, known as iPrEx, detailing how HIV-negative MSM at high risk for the virus could greatly reduce their risk of infection with the virus by taking a powder-blue tablet called Truvada (tenofovir disoproxil fumarate/emtricitabine). (Truvada, which contains a pair of ARVs, was approved in 2004 for use in combination with other ARVs to treat HIV.) Ultimately, researchers would determine that good adherence to the daily regimen among MSM lowered their chances of contracting HIV by an estimated 99 percent or more.
The Food and Drug Administration (FDA) approved PrEP for high-risk groups on July 16, 2012. But despite Truvada’s awesome potential to help send into retreat the scourge of HIV among MSM in particular—an estimated 70 percent of all new infections occur among this population—PrEP hit the scene with barely a whisper. Few sought out Truvada, and overall awareness remained minimal.
The tide began to turn in the fall of 2013 when a series of media outlets began asking why so few gay men were taking such a potentially transformative tool. The public attention helped open the floodgates, and the uptake of Truvada as prevention has been steadily rising ever since.
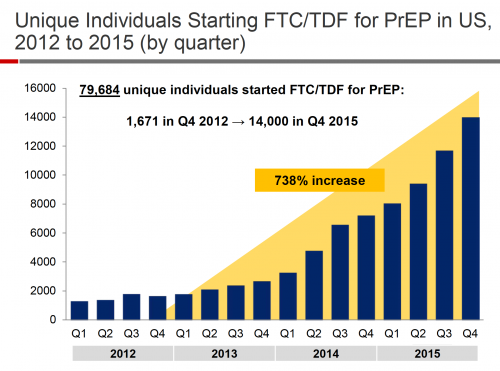
Gilead Sciences’ report on the estimated number of U.S. residents going on PrEP for the first time per quarter between 2012 and 2015. (FTC/TDF is shorthand for Truvada.) Note the steady increase launching in late 2013.Gilead Sciences
According to Gilead Sciences, which manufactures Truvada, an estimated 125,000 U.S. residents were on PrEP as of the first quarter of 2017. This is a remarkably robust figure given that a mere 22,000 were estimated to be on Truvada as prevention in early 2015.
Because of the long lag time in the CDC’s reporting about HIV diagnoses and estimated transmission rates—often the data are two to three years old by the time they’re made public—getting a firm handle on how PrEP has affected national HIV rates remains challenging.
A major question is whether PrEP is making it into the hands of those most at risk, where it can do the most good. Signs look promising. In 2015, the CDC estimated that 492,000 MSM (about a quarter of those 18 to 59 years old who don’t have HIV) are likely at high enough risk for HIV to be good candidates for PrEP. Meanwhile, data from Gilead suggest that the vast majority of PrEP users are MSM; and numerous other sources indicate that those who opt to go on PrEP do tend to be at higher risk for the virus. So it’s likely that an increasingly significant proportion of MSM at significant risk for HIV in the United States are indeed taking Truvada, positioning the drug to avert a considerable number of new infections.
PrEP use among MSM is particularly common in such cities as New York, San Francisco and Seattle, all of which have benefited from highly coordinated, localized efforts to promote PrEP among MSM.
San Francisco in particular has experienced a plummeting HIV diagnosis rate since 2012, a decline certainly tied to increasing treatment rates among the local HIV population, but also likely to PrEP as well. The Kaiser Permanente Northern California PrEP program, one of the largest in the nation, has data on a cumulative 5,000 years of PrEP use among its members. Thus far, there have been only a small handful of HIV transmissions among these individuals—cases limited to those whose PrEP use was interrupted because they experienced a lapse in insurance or temporarily dropped out of medical care with Kaiser. So despite initial signs in trials of PrEP that MSM would likely not adhere well to the daily drug regimen, this trove of real-world evidence strongly suggests otherwise.

San Francisco’s HIV epidemic stats between 2006 and 2015. Note the considerable drop in HIV diagnosis rates beginning in 2012.San Francisco Department of Health, HIV Epidemiology Report, Published Sept. 2016
In February, the CDC released an exciting estimate that overall U.S. HIV infection rates finally dropped 18 percent between 2008 and 2014. Researchers at the federal agency pointed to the effects of HIV treatment as prevention (TasP) as a likely major driver of the drop and speculated that PrEP may have affected the decline seen in 2014.
On the less promising side, the predominant users of PrEP appear to be white men over the age of 25. This means that Truvada use among those with the highest HIV risk—Black MSM, especially those under 25, maintain staggering rates of infection—remains particularly disappointing. These racial differences in PrEP uptake indicate that a tragic downside of the prevention pill is that it stands poised to widen existing racial disparities in HIV transmission rates among MSM.
Addressing such disparities, C. Bradley Hare, MD, the director of HIV care and prevention at Kaiser Permanente Medical Center in San Francisco, says “Clearly, we’re still missing very high-risk populations. We’re not penetrating into all the at-risk communities at the same levels we need to really drive down the epidemic across all the different risk groups.”
To follow is an overview of some of the most important scientific findings about PrEP, including details about its effectiveness, the three known cases of PrEP failure, and safety and side effects. There’s also the thorny, controversial question of how starting Truvada affects individuals’ level of sexual risk taking. And finally, we take a sneak peek at future forms of PrEP. Click on the hyperlinks for more about any of the research.

C. Bradley Hare addressing the 2016 Conference on Retroviruses and Opportunistic Infections in BostonCourtesy of Benjamin Ryan
How Effective Is PrEP?
There are two ways of examining how well PrEP works: on an individual level and on a population level. The placebo-controlled iPrEx trial found that the group of MSM who received PrEP had a 44 percent lower risk of HIV than those in the group that got the placebo. In 2015, researchers in the PROUD study in the United Kingdom—designed to better reflect real-world use of PrEP—announced that the group of MSM who received Truvada had an 86 percent lower HIV rate than those in the group assigned to wait and receive Truvada on a deferred basis.
Evidence reported in January 2017 suggested that the recent widespread practice among HIV-negative London MSM of purchasing a generic version of Truvada online—PrEP is not yet covered by the UK’s National Health Service (NHS)—was likely a key driver of a 40 percent one-year drop in HIV diagnosis rates at four London sexual health clinics that diagnose one third of the nation’s new cases of the virus.
As for how well PrEP works among individuals, its effectiveness is tied to how well people adhere to the daily regimen. For MSM, researchers have relied on mathematical modeling to estimate that taking Truvada daily is associated with a 99 percent HIV risk reduction, four to six pills weekly reduces risk by 96 percent, and two to three tablets weekly cuts risk by 76 percent.

Truvada tabletsIstock
Safety
Importantly, researchers have found that PrEP is not associated with any serious side effects, specifically known as grade 3 or 4 adverse events. Truvada use can give rise to two milder side effects in HIV-negative individuals, including small reductions in kidney function and bone mineral density.
Two decades of scientific research and clinical use inform clinicians’ knowledge of Truvada’s risks when used as treatment for HIV. But the research community still lacks long-term safety data for the tablet’s use among HIV-negative individuals because PrEP is relatively new on the scientific scene and the follow-up time of clinical trials is limited. In the meantime, one scientific paper reassuringly stated that short- or medium-term use of PrEP (up to five years) is as safe as aspirin.
Research indicates that PrEP-prompted changes in kidney function are generally reversible after an individual goes off Truvada. Additionally, Truvada has not apparently been responsible for any permanent kidney injury in HIV-negative individuals. And thanks to the recommended minimum of twice-yearly kidney function tests that go along with a PrEP prescription, clinicians can track any shifts that may be a cause for worry, in particular among older individuals and those with health conditions such as diabetes that may affect kidney health. (In two major PrEP studies, nearly all cases of creatinine clearance decline—an indication of reduced kidney function—occurred among those older than 40.)

PrEP may cause a modest decline in kidney function that is generally reversible after individuals stop taking Truvada.Istock
Thus far, Truvada use among HIV-negative individuals has not been linked to any bone fractures. And as with kidney function reduction, people who experience bone loss on PrEP will likely return to normal after stopping Truvada. Important questions do remain, however, about how Truvada may impede the natural bone growth that adolescents and young adults experience into their 20s and possibly lead to a lower bone density set point that could raise the risk of fractures later in life.
Research is currently under way to investigate ways to ward off bone problems among people on Truvada, such as through vitamin D or calcium supplements.
PrEP appears to pose a very low risk of prompting drug resistance to either of the drugs in Truvada. For such an effect to occur, an individual would need to contract the virus and then spend a period of time taking Truvada while HIV positive, allowing the virus to mutate in the presence of the medication. (The two drugs in Truvada are not sufficient to effectively treat an HIV infection; at least an additional third ARV is required.) In clinical trials of PrEP, only 0.5 percent of participants developed drug resistance, mostly because they contracted HIV shortly before starting PrEP (this group had a 37 percent chance of developing drug resistance), leading them to test false negative for the virus prior to going on Truvada.
Cases of PrEP Failure
Out of thousands of MSM who received PrEP in clinical trials, there were no cases of anyone acquiring HIV when tests indicated they were taking at least four tablets of Truvada per week. But the trials were not necessarily large enough to allow for what turned out to be such a rare outcome as a PrEP failure.
“If you looked at 10 times the number of people, might you have had a few failures?” says Kenneth Mayer, MD, medical research director at Fenway Health in Boston, an important locus of PrEP research and clinical practice. “We just don’t know.”
Thus far, in real-world practice there have been three documented cases of gay men contracting HIV while apparently adhering well to the daily PrEP regimen. The first two reported cases are simpler to make sense of. Both involved MSM contracting rare, highly drug-resistant strains of HIV that apparently evaded Truvada’s protective effects. Multiple sources of evidence supported each of these men’s claims that they were adherent to the daily Truvada regimen.
The third case involved a gay man in Amsterdam who was also adherent to PrEP according to multiple sources of evidence and who did not apparently contract a drug-resistant HIV strain. The researchers behind the PrEP study in which the man was a participant speculate that his “remarkably high” level of sexual risk taking—during his first three months on PrEP he had 90 sexual partners and engaged in 100 acts of condomless anal sex—led him to beat the odds in some way.

Elske Hoornenborg of the Public Health Service Amsterdam with her poster presentation on the Amsterdam PrEP failure case at CROI 2017 in SeattleBen Ryan
News of these three cases—the first story broke in February 2016—has caused a firestorm of interest among MSM. But despite the voluminous (and often acrimonious) nature of online chatter about these cases, they remain quite rare in the grand scope of widespread PrEP use. And while new cases will likely crop up with some regularity, there is every reason to believe that proportionally such PrEP failures will remain very uncommon.
That said, there is also reason to presume that other cases of PrEP failure have fallen below the radar. Each of the three known cases were reported by decidedly savvy clinicians or researchers who knew what steps to take, including the urgent need to order time-sensitive blood tests to ensure that a substantial level of scientific proof supported their claim to have discovered a case of true PrEP failure and not just an instance of HIV acquisition in the context of poor adherence to PrEP.
In Mayer’s words, the PrEP failure cases “show us that PrEP is not 100 percent protective.” However, he says, “I don’t feel like people should reflexively say you have to use condoms when you take PrEP. But I think for an informed consumer, if zero risk is key, then a ‘belt and suspenders’ make sense. And if STIs [sexually transmitted infections] are a concern, then again, condoms make sense.”
PrEP’s Role in Increased Sexual Risk Taking
PrEP remains most controversial because of how it may affect sexual risk taking among its users. Almost across the board, the trials of PrEP did not show that starting Truvada was associated with evidence of increased sexual risk taking—a phenomenon known as risk compensation—such as reported declines in condom use rates or increased STI diagnosis rates. However, a French trial of the non-daily-dosing regimen of PrEP reported in 2016 that when the participants switched from the placebo phase of the study to the open-label phase (when they all knew they were receiving Truvada), there was a significant drop in reported condom use. (For more information on what is sometimes known an intercourse-based PrEP protocol, click here.)

The French & Canadian IPERGAY study of an intercourse-based dosing protocol for PrEP saw a statistically significant rise in reported condomless sex rates after the double-blinded placebo phase ended and the open-label phase began.
As for real-world data on PrEP-associated risk compensation, such information is difficult to parse, given the multiple factors at play. Such moving parts include long declining rates of condom use among MSM, already-rising STI rates among MSM and the frequent STI testing that is a part of a PrEP prescription, which may play a role in driving up diagnosis rates.

STI rates have long been rising among MSM in particular.Centers for Disease Control and Prevention/2015 STD Surveillance Report
Voluminous anecdotal evidence has suggested a connection between PrEP use and changes in sexual behavior in the MSM community; those with a hookup app account in cities where PrEP is popular have likely witnessed considerable recent changes in personal safer-sex policies among fellow users. However, the major players in the PrEP research and clinical community, buttressed by PrEP’s clinical trial data, have long resisted acknowledging any such link. Their resistance is finally starting to soften as experiences with and analyses of real-world use of PrEP grow more detailed and nuanced.
Kaiser Permanente North California has provided the richest data regarding risk compensation effects among PrEP users. Researchers from the integrated health care system recently published a report on nearly 1,000 people who started PrEP through 2014 and were followed for a cumulative 850 years through June 2015. They found that quarterly rates of rectal gonorrhea and urethral chlamydia increased steadily in the group and approximately doubled after one year.
A survey of PrEP users at Kaiser’s San Francisco clinic, reported in late 2014, found that about four in 10 said they used condoms less after starting Truvada.

Fenway’s Kenneth MayerMarilyn Humphries
Mayer says he believes PrEP is contributing to increased sexual risk taking among MSM but does not believe Truvada use is the primary cause of such shifts.
“It’s safe to say that PrEP is definitely increasing [STI] screening rates, and it’s attracting a group of individuals who don’t want to use condoms,” Mayer says. “The question is, how much were they not using condoms in the past? That’s tricky. What I think is uncommon is somebody going into the clinic and saying, ‘I’ve used condoms every single day of my life up until now and today I’m ready to throw the condoms away.’”
“We’re certainly seeing increased high risk behavior, less condom use, and PrEP is one factor among others that is driving that, or contributing to that,” says Kaiser’s Hare.

A billboard promoting PrEP in West Hollywood points provocatively to Truvada’s capacity to lessen anxieties about sex.Courtesy of the Los Angeles LGBT Center
Whether or not PrEP causes a rise in STI rates, one thing is abundantly clear: PrEP use is associated with very high STI rates. (All the more reason those taking PrEP greatly benefit from the quarterly medical monitoring required to maintain a prescription and the recommended minimum of twice-yearly STI tests.) An analysis of MSM using PrEP in Seattle found they had chlamydia and gonorrhea rates more than 20 times that of the general MSM population. (The report also found men reported using condoms less for receptive anal intercourse during their first year on PrEP.) And a meta-analysis of numerous studies of MSM that reported on STIs found that the rates of new STI diagnoses among HIV-negative MSM on Truvada were 25 times greater for gonorrhea, 11 times greater for chlamydia and 45 times greater for syphilis, compared with the rates among MSM not on PrEP. Kaiser’s San Francisco clinic reported in September 2015 that half of all their patients on PrEP were diagnosed with at least one STI during their first year on Truvada.
New Forms of PrEP
When it comes to concerns about Truvada’s safety, efficacy or the ability of at-risk populations to use PrEP successfully, it’s important to note that the pale blue tablet is unlikely to remain the only player in the PrEP game for long. Two major clinical trials already under way are comparing new forms of PrEP with Truvada.
“We know that Truvada is not going to be the answer for everybody,” says Hare. “So we’re all looking for safer and different options for PrEP.”
One Phase III trial will determine whether Gilead’s Descovy (emtricitabine/tenofovir alafenamide) is as effective as Truvada in preventing HIV and whether it is associated with fewer side effects. (Three clinical trial phases are required before a treatment can gain approval.) Descovy contains an updated version of the tenofovir disoproxil fumarate component of Truvada that research in HIV-positive individuals indicates is safer for the bones and kidneys.

The FDA recently approved Descovy for use as a component of HIV treatment.
A second Phase III study is looking at a long-acting injectable form of PrEP: ViiV Healthcare’s ARV cabotegravir, given every eight weeks. Such a new PrEP modality could provide an excellent option to those who struggle with a daily drug regimen.
Additionally, a Phase II trial is investigating intravenous infusions of a broadly neutralizing antibody known as VRC01 for use as PrEP.
Results from the trials of Descovy, VRC01 and long-acting cabotegravir are expected in 2019, 2020 and 2021 respectively. Further down the road, we may see PrEP in the form of injectables requiring more widely spaced dosing, a subdermal implant and a microbicidal enema.
In the grand scope of the HIV epidemic, PrEP will ideally amount to a vital stopgap measure. For now, as the U.S. public health, advocacy and medical community strives to increase the proportion of the HIV population on successful ARVs, people at risk for the virus can greatly protect themselves by going on PrEP. Then, as the 2020s progress, the number of people who have untreated HIV and are therefore significantly infectious, will hopefully decline to such an extent that the need for PrEP will increasingly wane.
Benjamin Ryan is POZ’s editor at large, responsible for HIV science reporting. His work has also appeared in The New York Times, New York, The Nation, The Atlantic and The Marshall Project. Follow him on Facebook, Twitter and on his website, benryan.net.







5 Comments
5 Comments